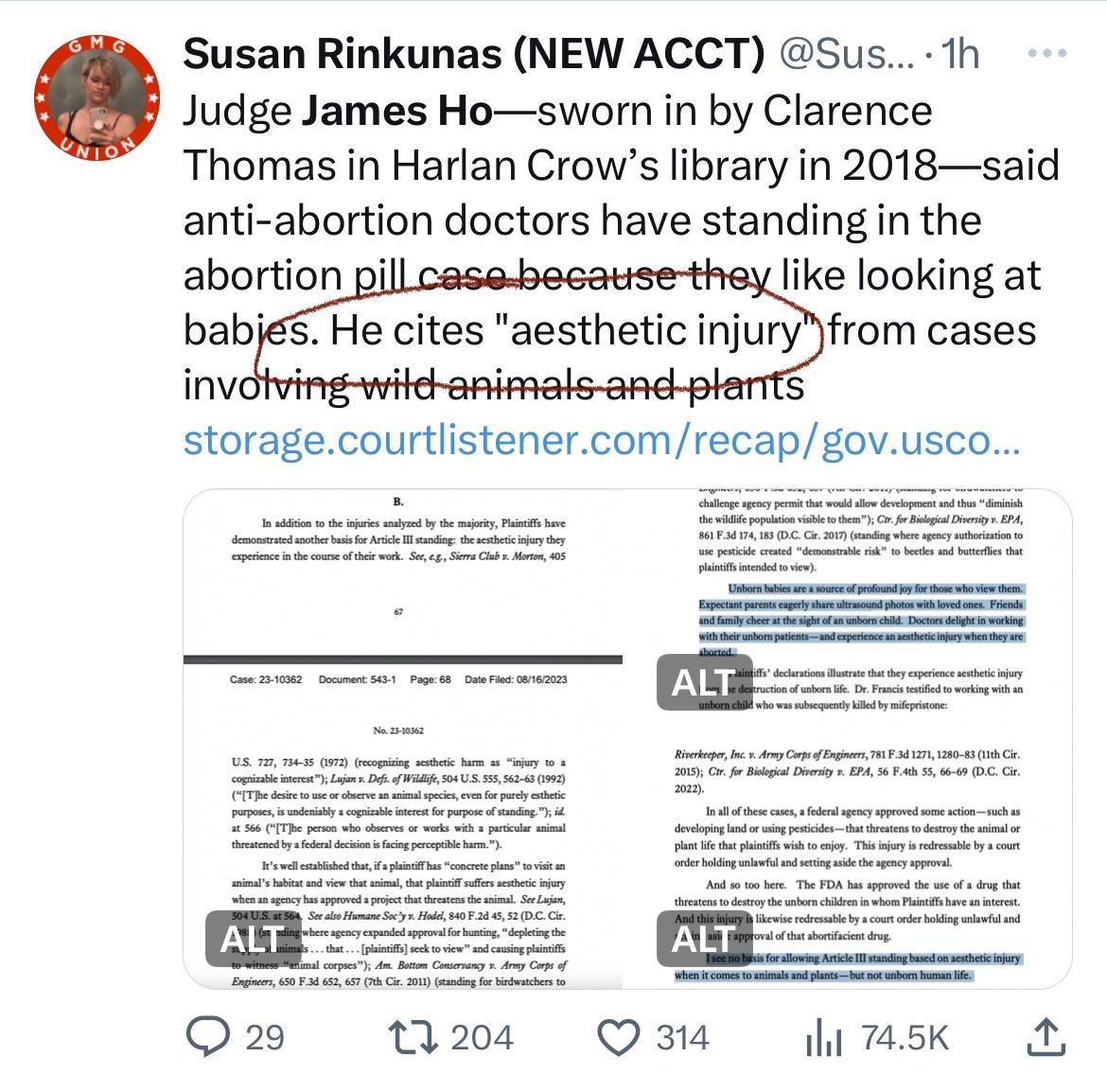
A federal appeals court on Wednesday said it would restrict access to a widely used abortion medication after finding that the federal government did not follow the proper process when it loosened regulations in 2016 to make the pill more easily available.
A three-judge panel of the conservative U.S. Court of Appeals for the 5th Circuit said Food and Drug Administration decisions to allow the drug mifepristone to be taken later in pregnancy, be mailed directly to patients and be prescribed by a medical professional other than a doctor were not lawful.
Despite the court’s ruling against the government and the drug manufacturer, mifepristone will remain available for now under existing regulations while the litigation continues, in accordance with a Supreme Court ruling this spring. Wednesday’s decision is likely to be appealed to the Supreme Court.
Skip to end of carousel
What is mifepristone?
Mifepristone is one of two drugs that work together to terminate a pregnancy, and is approved by the U.S. Food and Drug Administration through 10 weeks gestation. Mifepristone was first approved for use in France in 1988, and approved in the U.S. in 2000.
How a medication abortion works
A patient first takes mifepristone as a single pill, which blocks the hormone progesterone, preventing a pregnancy from progressing. About 24 hours later, the patient typically takes a four-pill dose of misoprostol to prompt contractions that expel the embryo or fetus.
Safety
A large body of research shows mifepristone is safe and effective. The American College of Obstetricians and Gynecologists analyzed hundreds of published studies, and found "serious side effects occur in less than 1% of patients, and major adverse events — significant infection, blood loss, or hospitalization — occur in less than 0.3%.”
Mifepristone and abortion access
Mifepristone is used in more than half of U.S. abortions, according to the Guttmacher Institute, a group that supports abortion rights. If mifepristone is taken off the market, abortion providers will have to provide only surgical abortions, or use a medication abortion regimen that includes only misoprostol.
Misoprostol-only medication abortion
A misoprostol-only abortion requires three doses of four pills each. While misoprostol is widely used on its own to perform abortions around the world, studies show it is less effective than the two-step regimen, and usually causes more cramping and potential side effects, including diarrhea, fever and chills.
1/5
End of carousel
Mifepristone is part of a two-drug regimen used in more than half of U.S. abortions and first approved for use in this country more than 20 years ago. The legal battle over the medication has intensified since the Supreme Court overturned Roe v. Wade’s grant of a constitutional right to abortion last June, a decision that spurred multiple states to further limit or ban the procedure.
The challenge to mifepristone was brought by the Alliance for Hippocratic Medicine, an association of antiabortion doctors and others. The group asserted that the FDA did not sufficiently consider safety concerns when it approved the drug or when it subsequently removed some restrictions — extending the approved use of mifepristone, for instance, through 10 weeks of pregnancy instead of seven.
The lawsuit, was filed in Amarillo, Tex., where U.S. District Judge Matthew Kacsmaryk — a Trump nominee with long-held antiabortion views — is the sole sitting judge. He issued an unprecedented ruling, for the first time suspending FDA approval of a human drug over objections from the agency. Kacsmaryk’s mifepristone opinion embraced language used by antiabortion activists, referring to abortion providers as “abortionists” and to fetuses and embryos as “unborn humans.”
A three-judge panel of the conservative U.S. Court of Appeals for the 5th Circuit said Food and Drug Administration decisions to allow the drug mifepristone to be taken later in pregnancy, be mailed directly to patients and be prescribed by a medical professional other than a doctor were not lawful.
Despite the court’s ruling against the government and the drug manufacturer, mifepristone will remain available for now under existing regulations while the litigation continues, in accordance with a Supreme Court ruling this spring. Wednesday’s decision is likely to be appealed to the Supreme Court.
Skip to end of carousel
How mifepristone is used
What is mifepristone?
Mifepristone is one of two drugs that work together to terminate a pregnancy, and is approved by the U.S. Food and Drug Administration through 10 weeks gestation. Mifepristone was first approved for use in France in 1988, and approved in the U.S. in 2000.
How a medication abortion works
A patient first takes mifepristone as a single pill, which blocks the hormone progesterone, preventing a pregnancy from progressing. About 24 hours later, the patient typically takes a four-pill dose of misoprostol to prompt contractions that expel the embryo or fetus.
Safety
A large body of research shows mifepristone is safe and effective. The American College of Obstetricians and Gynecologists analyzed hundreds of published studies, and found "serious side effects occur in less than 1% of patients, and major adverse events — significant infection, blood loss, or hospitalization — occur in less than 0.3%.”
Mifepristone and abortion access
Mifepristone is used in more than half of U.S. abortions, according to the Guttmacher Institute, a group that supports abortion rights. If mifepristone is taken off the market, abortion providers will have to provide only surgical abortions, or use a medication abortion regimen that includes only misoprostol.
Misoprostol-only medication abortion
A misoprostol-only abortion requires three doses of four pills each. While misoprostol is widely used on its own to perform abortions around the world, studies show it is less effective than the two-step regimen, and usually causes more cramping and potential side effects, including diarrhea, fever and chills.
1/5
End of carousel
Mifepristone is part of a two-drug regimen used in more than half of U.S. abortions and first approved for use in this country more than 20 years ago. The legal battle over the medication has intensified since the Supreme Court overturned Roe v. Wade’s grant of a constitutional right to abortion last June, a decision that spurred multiple states to further limit or ban the procedure.
The challenge to mifepristone was brought by the Alliance for Hippocratic Medicine, an association of antiabortion doctors and others. The group asserted that the FDA did not sufficiently consider safety concerns when it approved the drug or when it subsequently removed some restrictions — extending the approved use of mifepristone, for instance, through 10 weeks of pregnancy instead of seven.
The lawsuit, was filed in Amarillo, Tex., where U.S. District Judge Matthew Kacsmaryk — a Trump nominee with long-held antiabortion views — is the sole sitting judge. He issued an unprecedented ruling, for the first time suspending FDA approval of a human drug over objections from the agency. Kacsmaryk’s mifepristone opinion embraced language used by antiabortion activists, referring to abortion providers as “abortionists” and to fetuses and embryos as “unborn humans.”