The Justice Department on Monday appealed a Texas judge’s decision that would block access to a key abortion drug across the country, arguing that the challengers had no right to file the lawsuit since they were not personally harmed by the abortion pill.
The appeal, filed in the right-leaning U.S. Court of Appeals for the 5th Circuit, landed less than one business day after Judge Matthew Kascmaryk on Friday evening ordered the Federal Drug Administration to revoke its approval of mifepristone — one of the two medications used in more than half of all abortions in the United States.
Kascmaryk had said his ruling would not take effect until this Friday, giving the Biden administration time to appeal before the drug could be pulled from the marketplace. In its filing Monday, the government asked the 5th Circuit judges to keep Kascmaryk’s order on hold until the appeal is decided.
Kacsmaryk’s ruling, Justice Department lawyers said, had “upended decades of reliance by blocking FDA’s approval of mifepristone and depriving patients of access to this safe and effective treatment, based on the court’s own misguided assessment of the drug’s safety.”
The government asked the appeals court to issue its decision on pausing Kacsmaryk’s order by Thursday at noon.
If the appeals court does not pause, or stay, the order while the appeal is decided, or is slow in making that decision, the Justice Department could go directly to the U.S. Supreme Court in an effort to keep mifepristone available for women seeking to terminate their pregnancies. The high court will almost certainly hear the case eventually, either before or after the 5th Circuit rules.
The Justice Department and the company that makes mifepristone said in their appeal that the antiabortion groups challenging the drug lack sufficient legal grounds or standing to proceed with their lawsuit.
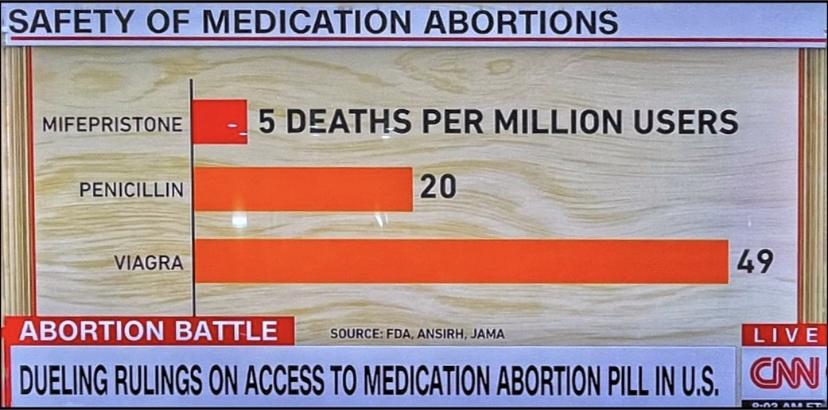
To establish standing, the groups have to demonstrate that they face a risk of injury. The Justice Department characterized as speculative the doctors’ claims that they are directly harmed because their patients claim to have experienced complications from the medication. Mifepristone was approved 23 years ago after clinical trials including thousands of pregnant patients showed a low rate of complications.
“Plaintiffs lack standing to challenge FDA’s approval of a drug they neither take nor prescribe; their challenge to FDA actions dating back to 2000 is manifestly untimely; and they have provided no basis for second-guessing FDA’s scientific judgment,” the appeal said.
In a divided nation, dueling decisions on the abortion pill
Both antiabortion and abortion rights advocates view Texas lawsuit as a landmark case that would shape abortion access in the country and could ultimately present Supreme Court justices with their biggest test on the issue since their decision in June to overturn the guarantee of abortion rights provided by Roe v. Wade.
In November, conservative groups filed a lawsuit in Kascmaryk’s Amarillo, Tex., courthouse seeking to reverse the FDA’s approval of mifepristone as safe and effective, including in states where abortion rights are protected.
Kascmaryk, a lifelong antiabortion advocate is the sole judge in his district and has been a go-to judge for conservatives seeking to challenge Biden administration policies.
In a four-hour hearing last month, the attorneys for the conservative group argued that the pill is unsafe — a claim that has been sharply refuted by medical experts — and that the FDA did not follow proper protocol when approving its use.
The government’s attorneys argued that the scientific evidence has repeatedly proven mifepristone to be safe and effective since the FDA approved its use more than 20 years ago.
Both groups acknowledged in the hearing that there is no precedent for a court to order the suspension of a long-approved medication.
Separately, a group of Democratic state attorneys general filed a lawsuit in February in Washington state, seeking to expand access to mifepristone.
Shortly after Kasmaryk issued his ruling on Friday, Judge Thomas O. Rice delivered his ruling in the Washington state case, ordering the the FDA to preserve “the status quo” and retain access to the abortion medication in the 17 states — along with D.C. — that filed the lawsuit.
Even the Justice Department was somewhat unclear about the path forward, given the two conflicting rulings. On Monday, the Justice Department asked the judge in the Washington state case for clarification for how to reconcile the contradictory opinions.
The appeal, filed in the right-leaning U.S. Court of Appeals for the 5th Circuit, landed less than one business day after Judge Matthew Kascmaryk on Friday evening ordered the Federal Drug Administration to revoke its approval of mifepristone — one of the two medications used in more than half of all abortions in the United States.
Kascmaryk had said his ruling would not take effect until this Friday, giving the Biden administration time to appeal before the drug could be pulled from the marketplace. In its filing Monday, the government asked the 5th Circuit judges to keep Kascmaryk’s order on hold until the appeal is decided.
Kacsmaryk’s ruling, Justice Department lawyers said, had “upended decades of reliance by blocking FDA’s approval of mifepristone and depriving patients of access to this safe and effective treatment, based on the court’s own misguided assessment of the drug’s safety.”
The government asked the appeals court to issue its decision on pausing Kacsmaryk’s order by Thursday at noon.
If the appeals court does not pause, or stay, the order while the appeal is decided, or is slow in making that decision, the Justice Department could go directly to the U.S. Supreme Court in an effort to keep mifepristone available for women seeking to terminate their pregnancies. The high court will almost certainly hear the case eventually, either before or after the 5th Circuit rules.
The Justice Department and the company that makes mifepristone said in their appeal that the antiabortion groups challenging the drug lack sufficient legal grounds or standing to proceed with their lawsuit.
To establish standing, the groups have to demonstrate that they face a risk of injury. The Justice Department characterized as speculative the doctors’ claims that they are directly harmed because their patients claim to have experienced complications from the medication. Mifepristone was approved 23 years ago after clinical trials including thousands of pregnant patients showed a low rate of complications.
“Plaintiffs lack standing to challenge FDA’s approval of a drug they neither take nor prescribe; their challenge to FDA actions dating back to 2000 is manifestly untimely; and they have provided no basis for second-guessing FDA’s scientific judgment,” the appeal said.
In a divided nation, dueling decisions on the abortion pill
Both antiabortion and abortion rights advocates view Texas lawsuit as a landmark case that would shape abortion access in the country and could ultimately present Supreme Court justices with their biggest test on the issue since their decision in June to overturn the guarantee of abortion rights provided by Roe v. Wade.
In November, conservative groups filed a lawsuit in Kascmaryk’s Amarillo, Tex., courthouse seeking to reverse the FDA’s approval of mifepristone as safe and effective, including in states where abortion rights are protected.
Kascmaryk, a lifelong antiabortion advocate is the sole judge in his district and has been a go-to judge for conservatives seeking to challenge Biden administration policies.
In a four-hour hearing last month, the attorneys for the conservative group argued that the pill is unsafe — a claim that has been sharply refuted by medical experts — and that the FDA did not follow proper protocol when approving its use.
The government’s attorneys argued that the scientific evidence has repeatedly proven mifepristone to be safe and effective since the FDA approved its use more than 20 years ago.
Both groups acknowledged in the hearing that there is no precedent for a court to order the suspension of a long-approved medication.
Separately, a group of Democratic state attorneys general filed a lawsuit in February in Washington state, seeking to expand access to mifepristone.
Shortly after Kasmaryk issued his ruling on Friday, Judge Thomas O. Rice delivered his ruling in the Washington state case, ordering the the FDA to preserve “the status quo” and retain access to the abortion medication in the 17 states — along with D.C. — that filed the lawsuit.
Even the Justice Department was somewhat unclear about the path forward, given the two conflicting rulings. On Monday, the Justice Department asked the judge in the Washington state case for clarification for how to reconcile the contradictory opinions.