Meanwhile....back in RealityLand (and loads of Cochrane-related sources, considered the Gold Standard for research)
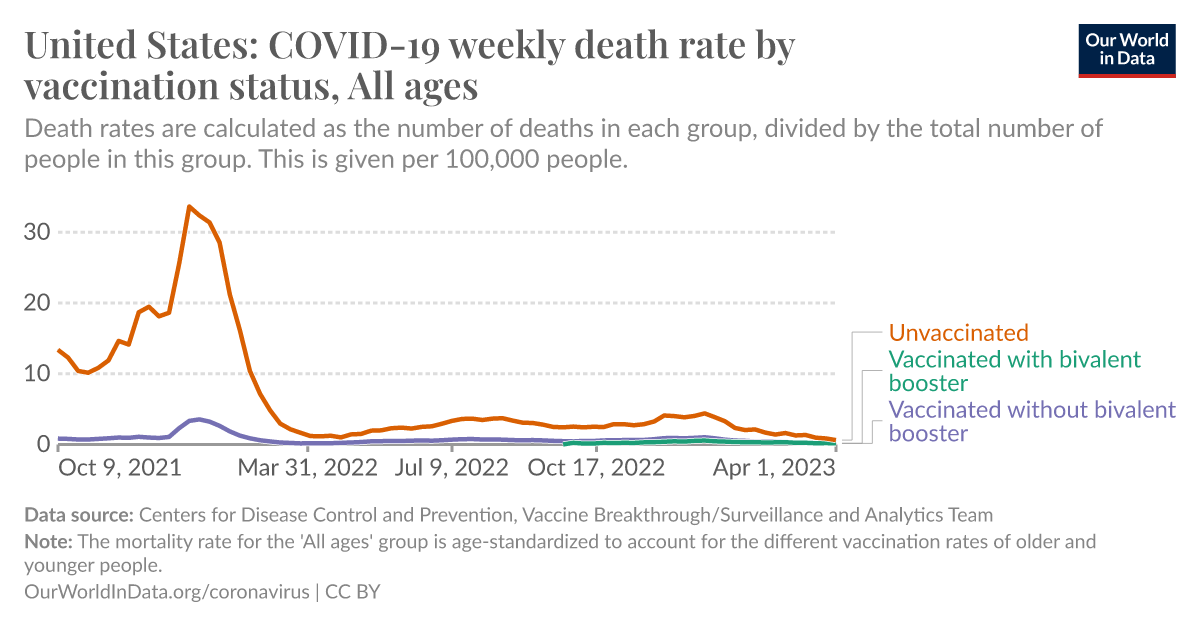
Compared to placebo, most vaccines reduce, or likely reduce, the proportion of participants with confirmed symptomatic COVID-19, and for some, there is high-certainty evidence that they reduce severe or critical disease. There is probably little or no difference between most vaccines and placebo...

pubmed.ncbi.nlm.nih.gov
Authors' conclusions: Compared to placebo, most vaccines reduce, or likely reduce, the proportion of participants with confirmed symptomatic COVID-19, and for some, there is high-certainty evidence that they reduce severe or critical disease. There is probably little or no difference between most vaccines and placebo for serious adverse events. Over 300 registered RCTs are evaluating the efficacy of COVID-19 vaccines, and this review is updated regularly on the COVID-NMA platform (covid-nma.com). Implications for practice Due to the trial exclusions, these results cannot be generalized to pregnant women, individuals with a history of SARS-CoV-2 infection, or immunocompromized people. Most trials had a short follow-up and were conducted before the emergence of variants of concern. Implications for research Future research should evaluate the long-term effect of vaccines, compare different vaccines and vaccine schedules, assess vaccine efficacy and safety in specific populations, and include outcomes such as preventing long COVID-19. Ongoing evaluation of vaccine efficacy and effectiveness against emerging variants of concern is also vital.
Affiliations
- 1Cochrane France, Paris, France.
- 2Centre of Research in Epidemiology and Statistics (CRESS), INSERM, INRAE, Université de Paris, Paris, France.
- 3Cochrane Review Group on Drugs and Alcohol, Rome, Italy.
- 4Cochrane Response, Cochrane, London, UK.
- 5Department of Immunization, Vaccines and Biologicals, World Health Organization, Geneva, Switzerland.
- 6Evidence Synthesis Ireland, Cochrane Ireland and HRB-Trials Methodology Research Network, National University of Ireland, Galway, Ireland.
- 7UCD Centre for Experimental Pathogen Host Research and UCD School of Medicine, University College Dublin, Dublin, Ireland.
- 8Department of Clinical Immunology and Infectious Diseases, Henri Mondor Hospital, Vaccine Research Institute, Université Paris Est Créteil, Paris, France.
- 9Quality Assurance Norms and Standards Department, World Health Organization, Geneva, Switzerland.
- 10Cochrane South Africa, South African Medical Research Council, Cape Town, South Africa.
- 11Institute for Evidence in Medicine, Medical Center & Faculty of Medicine, University of Freiburg, Freiburg, Germany.
- 12Cochrane Germany, Cochrane Germany Foundation, Freiburg, Germany.
- 13Department of Anesthesia, Intensive Care and Emergency, Fondazione IRCCS Ca' Granda Ospedale Maggiore Policlinico, Department of Pathophysiology and Transplantation, University of Milan, Milan, Italy.
- 14Epistemonikos Foundation, Santiago, Chile.
- 15UC Evidence Center, Cochrane Chile Associated Center, Pontificia Universidad Católica de Chile, Santiago, Chile.
- 16Centre for Evidence Based Medicine Odense (CEBMO) and Cochrane Denmark, University of Southern Denmark, Odense, Denmark.
- 17Open Patient data Explorative Network (OPEN), Odense University Hospital, Odense, Denmark.
Conflict of interest statement
Carolina Graña: none known.
Lina Ghosn: none known.
Theodoros Evrenoglou: none known.
Alexander Jarde: none known.
Silvia Minozzi: no relevant interests; Joint Co‐ordinating Editor and Method editor of the Drugs and Alcohol Group.
Hanna Bergman: Cochrane Response – consultant; WHO – grant/contract (Cochrane Response was commissioned by the WHO to perform review tasks that contribute to this publication).
Brian Buckley: none known.
Katrin Probyn: Cochrane Response – consultant; WHO – consultant (Cochrane Response was commissioned to perform review tasks that contribute to this publication).
Gemma Villanueva: Cochrane Response – employment (Cochrane Response has been commissioned by WHO to perform parts of this systematic review).
Nicholas Henschke: Cochrane Response – consultant; WHO – consultant (Cochrane Response was commissioned by the WHO to perform review tasks that contributed to this publication).
Hillary Bonnet: none known.
Rouba Assi: none known.
Sonia Menon: P95 – consultant.
Melanie Marti: no relevant interests; Medical Officer at WHO.
Declan Devane: Health Research Board (HRB) – grant/contract; registered nurse and registered midwife but no longer in clinical practice; Editor, Cochrane Pregnancy and Childbirth Group.
Patrick Mallon: AstraZeneca – Advisory Board; spoken of vaccine effectiveness to media (print, online, and live); works as a consultant in a hospital that provides vaccinations; employed by St Vincent's University Hospital.
Jean‐Daniel Lelievre: no relevant interests; published numerous interviews in the national press on the subject of COVID vaccination; Head of the Department of Infectious Diseases and Clinical Immunology CHU Henri Mondor APHP, Créteil; WHO (IVRI‐AC): expert Vaccelarate (European project on COVID19 Vaccine): head of WP; involved with COVICOMPARE P et M Studies (APHP, INSERM) (public fundings).
Lisa Askie: no relevant interests; Co‐convenor, Cochrane Prospective Meta‐analysis Methods Group.
Tamara Kredo: no relevant interests; Medical Officer in an Infectious Diseases Clinic at Tygerberg Hospital, Stellenbosch University.
Gabriel Ferrand: none known.
Mauricia Davidson: none known.
Carolina Riveros: no relevant interests; works as an epidemiologist.
David Tovey: no relevant interests; Emeritus Editor in Chief, Feedback Editors for 2 Cochrane review groups.
Joerg J Meerpohl: no relevant interests; member of the German Standing Vaccination Committee (STIKO).
Giacomo Grasselli: Pfizer – speaking engagement.
Gabriel Rada: none known.
Asbjørn Hróbjartsson: no relevant interests; Cochrane Methodology Review Group Editor.
Philippe Ravaud: no relevant interests; involved with Mariette CORIMUNO‐19 Collaborative 2021, the Ministry of Health, Programme Hospitalier de Recherche Clinique, Foundation for Medical Research, and AP‐HP Foundation.
Anna Chaimani: none known.
Isabelle Boutron: no relevant interests; member of Cochrane Editorial Board.
Awful lot of "no conflicts" listed there....